This post consults a number of published articles and lifts pertinent ideas from them without going into great depth. From these excerpts, we can see how much progress has been made in only the past couple of years, and the directions in which future research will proceed.
A report published by the International Journal of Obesity begins by noting that visceral fat is associated with increased inflammation, and also with increased risk of obesity-related co-morbidities. What has a high-fat diet to do with all this? The subjects of this study were obesity-prone (OP) rats and obesity-resistant (OR) rats. Both types of lab rodents were divided into groups that received either low-fat or high-fat diets, and the researchers looked for inflammatory markers in their adipose tissue.
The Conclusions paragraph says that “a higher susceptibility to developing obesity is characterized by large adipocytes and increased visceral adipose inflammation.” Even in the OR rats, visceral adipose inflammation showed up secondary to even small weight increases, which the scientists read as a strong hint that their vulnerability to obesity-related co-morbidities would also increase.
Macrophages are immune cells that regulate local metabolism, and are supposed to clean up the debris from damaged tissue and take steps toward repair. Sometimes they get out of hand, and that is a problem.
They are also malleable, since stimuli from within or without can polarize them toward phenotypes with distinct functions. Here is where it gets interesting:
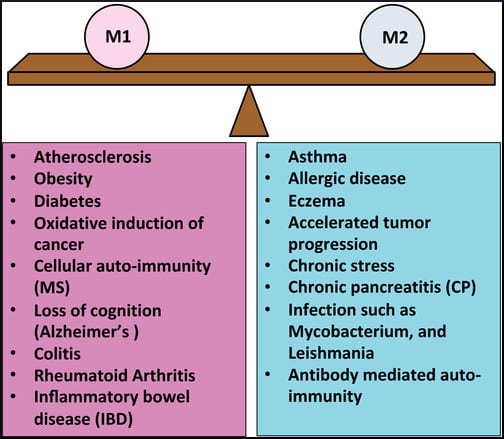
M1 and M2 macrophages may coexist in different organs and may differentially affect asthma and obesity… While M1 macrophages are considered beneficial in asthma and contribute to the pathology of obesity, M2 macrophages contribute to the pathology of asthma, but limit metabolic syndrome associated with obesity.
In adipose tissue (fat), most immune cells are macrophages. If a person is lean, they may account for less than 10% of the fat tissue cells, while in an extremely obese person, their population might be more than 50%.
Another article defines obesity as “a chronic low-grade inflammatory state” and looks into the question of what could alter macrophage polarization. The answer is metformin, which decreases the liver’s glucose production and increases insulin sensitivity, and is the drug most widely prescribed for type 2 diabetes. It improves the chronic inflammatory state, but the “why” is still up for grabs.
Another class of drugs, the Thiazolidinediones (or TZDs) also counter insulin resistance by riding herd on the macrophages, “controlling the metabolic fuel they use to generate energy.” These pharmaceuticals may limit the damage done by tissue inflammation, and the article describing their work explains things in yet another set of words:
Overnutrition […] causes a buildup of fat that can significantly damage tissues. When this happens, macrophages infiltrate the affected tissues, sequester free fatty acids, and help repair damaged tissue — essentially acting as a protector of the body during times of metabolic stress. However, extended stress on these tissues activates inflammatory characteristics in macrophages that contribute to several systemic effects of obesity including diabetes, atherosclerosis, and cardiovascular disease.
Your responses and feedback are welcome!
Source: “High fat diet consumption differentially affects adipose tissue inflammation and adipocyte size in obesity-prone and obesity-resistant rats,” Nature.com, 11/20/17
Source: “Macrophages—common culprit in obesity and asthma,” Wiley.com, 11/27/17
Source: “Metformin improves obesity-associated inflammation by altering macrophages polarization,” ScienceDirect.com, 02/05/18
Source: “Diabetes drugs act as powerful curb for immune cells in controlling inflammation,” ScienceDaily.com, 07/28/18
Image source: Public Domain

 FAQs and Media Requests:
FAQs and Media Requests: 












One Response